Matrix reforming of hydrocarbons: New possibilities for low-tonnage gas processing and energy
DOI:
https://doi.org/10.18686/cest537Keywords:
natural gas; energy; gas chemistry; oxidative conversion; matrix reforming; syngas; hydrogenAbstract
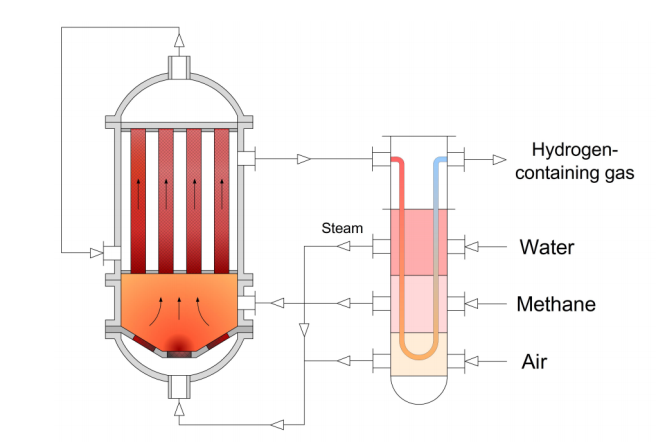
The huge unconventional resources of natural gas in the earth's crust make it, in the future, not only the main feedstock for global energy, but also the cheapest and most abundant raw material for the production of many basic petrochemical products. However, technological complexity and high energy consumption for modern multistage processes of converting methane into thermodynamically less stable products remain the main problem constraining the development of gas chemistry. Currently, less complex and more flexible non-catalytic conversion processes of hydrocarbon gases are of great interest. A promising solution may be a principally new technology of autothermal matrix reforming of natural gas into syngas or hydrogen. Matrix reforming is a type of non-catalytic partial oxidation of natural gas with internal recuperation of the heat of conversion products, which is implemented in the surface combustion mode. It is an autothermal process that does not require additional heat supply. The absence of a catalyst not only simplifies the process but also dramatically reduces the requirements for gas preparation and purification, making the process insensitive to many impurities that are catalytic poisons and allowing the direct use of hydrocarbon gases of almost any composition as feedstock, including associated and refinery gases, as well as low-boiling liquid hydrocarbons up to the kerosene fraction. The paper presents the basic principles of matrix reforming and its kinetics, its advantages, the results achieved so far, and the most promising areas of its application.
Downloads
Published
How to Cite
Issue
Section
License
Copyright (c) 2026 Vladimir Arutyunov, Valery Savchenko, Aleksey Nikitin, Igor Sedov

This work is licensed under a Creative Commons Attribution 4.0 International License.
References
1. Aasberg-Petersen K, Dybkjær I, Ovesen CV, et al. Natural gas to synthesis gas – Catalysts and catalytic processes. Journal of Natural Gas Science and Engineering. 2011; 3(2): 423–459. doi: 10.1016/j.jngse.2011.03.004 DOI: https://doi.org/10.1016/j.jngse.2011.03.004
2. Dybkjær I, Aasberg‐Petersen K. Synthesis gas technology large‐scale applications. The Canadian Journal of Chemical Engineering. 2016; 94(4): 607–612. doi: 10.1002/cjce.22453 DOI: https://doi.org/10.1002/cjce.22453
3. Speight JG. Handbook of Industrial Hydrocarbon Processes, 2nd ed. Gulf Professional Publishing; 2020.
4. Szablowski L, Wojcik M, Dybinski O. Review of steam methane reforming as a method of hydrogen production. Energy. 2025; 316: 134540. doi: 10.1016/j.energy.2025.134540 DOI: https://doi.org/10.1016/j.energy.2025.134540
5. Aasberg-Petersen K, Bak Hansen JH, Christensen TS, et al. Technologies for large-scale gas conversion. Applied Catalysis A: General. 2001; 221(1–2): 379–387. doi: 10.1016/S0926-860X(01)00811-0 DOI: https://doi.org/10.1016/S0926-860X(01)00811-0
6. Iglesia E, Spivey JJ, Fleisch TH. Natural Gas Conversion VI. Elsevier; 2001. doi: 10.1016/S0167-2991(01)X8267-0 DOI: https://doi.org/10.1016/S0167-2991(01)X8267-0
7. Chen CM, Bennett DL, Carolan MF, et al. ITM Syngas ceramic membrane technology for synthesis gas production. In: Studies in Surface Science and Catalysis. Elsevier; 2004. pp. 55–60. doi: 10.1016/S0167-2991(04)80027-2 DOI: https://doi.org/10.1016/S0167-2991(04)80027-2
8. Mazanec T, Perry S, Tonkovich L, et al. Microchannel gas-to-liquids conversion-thinking big by thinking small. In: Studies in Surface Science and Catalysis. Elsevier; 2004. pp. 169–174. doi: 10.1016/S0167-2991(04)80046-6 DOI: https://doi.org/10.1016/S0167-2991(04)80046-6
9. CompactGTL. Available online: https://www.cbinsights.com/company/compactgtl (accessed on 15 January 2025).
10. Zhang L, Wu W, Zhang Y, et al. Clean synthesis gas production from municipal solid waste via catalytic gasification and reforming technology. Catalysis Today. 2018; 318: 39–45. doi: 10.1016/j.cattod.2018.02.050 DOI: https://doi.org/10.1016/j.cattod.2018.02.050
11. Zhang H, Sun Z, Hu YH. Steam reforming of methane: Current states of catalyst design and process upgrading. Renewable and Sustainable Energy Reviews. 2021; 149: 111330. doi: 10.1016/j.rser.2021.111330 DOI: https://doi.org/10.1016/j.rser.2021.111330
12. Savchenko VI, Nikitin AV, Zimin YS, et al. Impact of post-flame processes on the hydrogen yield in partial oxidation of methane in the matrix reformer. Chemical Engineering Research and Design. 2021; 175: 250–258. doi: 10.1016/j.cherd.2021.09.009 DOI: https://doi.org/10.1016/j.cherd.2021.09.009
13. Eastman D. Synthesis Gas by Partial Oxidation. Industrial & Engineering Chemistry. 1956; 48(7): 1118–1122. doi: 10.1021/ie50559a016 DOI: https://doi.org/10.1021/ie50559a016
14. Kuhre CJ, Shearer CJ. Partial oxidation grows stronger in U.S. The Oil and Gas Journal. 1971; 85–90.
15. Dahl PJ, Christensen TS, Winter-Madsen S, et al. Proven autothermal reforming technology for modern large-scale methanol plants. In: Proceedings of the Nitrogen + Syngas International Conference & Exibition; 24–27 February 2014; Paris, France.
16. Zhang Q, Liu Y, Chen T, et al. Simulations of methane partial oxidation by CFD coupled with detailed chemistry at industrial operating conditions. Chemical Engineering Science. 2016; 142: 126–136. doi: 10.1016/j.ces.2015.11.010 DOI: https://doi.org/10.1016/j.ces.2015.11.010
17. Chen D, Chen X, Luo C, et al. Structural design and performance evaluation of industrial-scale C2H2 reactor by partial oxidation of natural gas. Chemical Engineering Journal. 2021; 426: 130871. doi: 10.1016/j.cej.2021.130871 DOI: https://doi.org/10.1016/j.cej.2021.130871
18. Liu Y, Zhang Q, Wang T. Detailed Chemistry Modeling of Partial Combustion of Natural Gas for Coproducing Acetylene and Syngas. Combustion Science and Technology. 2017; 189(5): 908–922. doi: 10.1080/00102202.2016.1256879 DOI: https://doi.org/10.1080/00102202.2016.1256879
19. Bodke AS, Olschki DA, Schmidt LD, et al. High Selectivities to Ethylene by Partial Oxidation of Ethane. Science. 1999; 285(5428): 712–715. doi: 10.1126/science.285.5428.712 DOI: https://doi.org/10.1126/science.285.5428.712
20. Schmidt LD. Modeling Millisecond Reactors. In: Studies in Surface Science and Catalysis. Elsevier; 2001. pp. 1–12. doi: 10.1016/S0167-2991(01)80272-X DOI: https://doi.org/10.1016/S0167-2991(01)80272-X
21. Basini LE, Guarinoni A. Short Contact Time Catalytic Partial Oxidation (SCT-CPO) for Synthesis Gas Processes and Olefins Production. Industrial & Engineering Chemistry Research. 2013; 52(48): 17023–17037. doi: 10.1021/ie402463m DOI: https://doi.org/10.1021/ie402463m
22. Al-Hamamre Z, Al-Zoubi A. The use of inert porous media based reactors for hydrogen production. International Journal of Hydrogen Energy. 2010; 35(5): 1971–1986. doi: 10.1016/j.ijhydene.2009.11.079 DOI: https://doi.org/10.1016/j.ijhydene.2009.11.079
23. Nourbakhsh H, Rahbar Shahrouzi J, Ebrahimi H, et al. Experimental and numerical study of syngas production during premixed and ultra-rich partial oxidation of methane in a porous reactor. International Journal of Hydrogen Energy. 2019; 44(60): 31757–31771. doi: 10.1016/j.ijhydene.2019.10.084 DOI: https://doi.org/10.1016/j.ijhydene.2019.10.084
24. Toledo M, Bubnovich V, Saveliev A, et al. Hydrogen production in ultrarich combustion of hydrocarbon fuels in porous media. International Journal of Hydrogen Energy. 2009; 34(4): 1818–1827. doi: 10.1016/j.ijhydene.2008.12.001 DOI: https://doi.org/10.1016/j.ijhydene.2008.12.001
25. Toledo MG, Utria KS, González FA, et al. Hybrid filtration combustion of natural gas and coal. International Journal of Hydrogen Energy. 2012; 37(8): 6942–6948. doi: 10.1016/j.ijhydene.2012.01.061 DOI: https://doi.org/10.1016/j.ijhydene.2012.01.061
26. Dorofeenko SO, Polianczyk EV. Conversion of hydrocarbon gases to synthesis gas in a reversed-flow filtration combustion reactor. Chemical Engineering Journal. 2016; 292: 183–189. doi: 10.1016/j.cej.2016.02.013 DOI: https://doi.org/10.1016/j.cej.2016.02.013
27. Dorofeenko SO, Polianczyk EV, Tsvetkov MV. Toward the ultimate efficiency of methane to syngas conversion by partial oxidation: A moving bed reactor with parallel preheating of reactants. Fuel. 2024; 363: 131005. doi: 10.1016/j.fuel.2024.131005 DOI: https://doi.org/10.1016/j.fuel.2024.131005
28. Closed Joint-Stock Company Rusinvest. Synthesis gas production process. Patent RU2136580, 10 September 1999.
29. Lim EG, Dames EE, Cedrone KD, et al. The engine reformer: Syngas production in an engine for compact gas‐to‐liquids synthesis. The Canadian Journal of Chemical Engineering. 2016; 94(4): 623–635. doi: 10.1002/cjce.22443 DOI: https://doi.org/10.1002/cjce.22443
30. Cho M, Oh W, Park N, et al. Experimental study on the effects of gas additives on uncatalyzed partial oxidation of methane in a spark-ignition engine. International Journal of Engine Research. 2025; 26(2): 149–160. doi: 10.1177/14680874241272862 DOI: https://doi.org/10.1177/14680874241272862
31. Cornelissen R, Tober E, Kok J, et al. Generation of synthesis gas by partial oxidation of natural gas in a gas turbine. Energy. 2006; 31(15): 3199–3207. doi: 10.1016/j.energy.2006.03.028 DOI: https://doi.org/10.1016/j.energy.2006.03.028
32. Granovskiy M. Integrated Coproduction of Power and Syngas from Natural Gas to Abate Greenhouse Gas Emissions without Economic Penalties. ACS Omega. 2021; 6(25): 16336–16342. doi: 10.1021/acsomega.1c00743 DOI: https://doi.org/10.1021/acsomega.1c00743
33. Buravtsev NN, Kolbanovskii YA, Rossikhin IV, et al. Effect of the Calorific Intensity of Combustion Chamber on Production of Synthesis Gas in Partial Oxidation of Methane–Oxygen Mixtures in the Combustion Mode. Russian Journal of Applied Chemistry. 2018; 91(10): 1588–1596. doi: 10.1134/S107042721810004X DOI: https://doi.org/10.1134/S107042721810004X
34. Levikhin AA, Boryaev AA. High-temperature reactor for hydrogen production by partial oxidation of hydrocarbons. International Journal of Hydrogen Energy. 2023; 48(72): 28187–28204. doi: 10.1016/j.ijhydene.2023.03.459 DOI: https://doi.org/10.1016/j.ijhydene.2023.03.459
35. Aasberg-Petersen K, Christensen TS, Dybkjaer I, et al. Synthesis gas production for FT synthesis. In: Studies in Surface Science and Catalysis. Elsevier; 2004. pp. 258–405. doi: 10.1016/S0167-2991(04)80461-0 DOI: https://doi.org/10.1016/S0167-2991(04)80461-0
36. Soleimani S, Lehner M. Tri-Reforming of Methane: Thermodynamics, Operating Conditions, Reactor Technology and Efficiency Evaluation—A Review. Energies. 2022; 15(19): 7159. doi: 10.3390/en15197159 DOI: https://doi.org/10.3390/en15197159
37. Nikitin A, Ozersky A, Savchenko V, et al. Matrix conversion of natural gas to syngas: The main parameters of the process and possible applications. Chemical Engineering Journal. 2019; 377: 120883. doi: 10.1016/j.cej.2019.01.162 DOI: https://doi.org/10.1016/j.cej.2019.01.162
38. Savchenko VI, Zimin YS, Nikitin AV, et al. Utilization of CO2 in non-catalytic dry reforming of C1–C4 hydrocarbons. Journal of CO2 Utilization. 2021; 47: 101490. doi: 10.1016/j.jcou.2021.101490 DOI: https://doi.org/10.1016/j.jcou.2021.101490
39. Healy D, Kalitan DM, Aul CJ, et al. Oxidation of C1−C5 Alkane Quinternary Natural Gas Mixtures at High Pressures. Energy & Fuels. 2010; 24(3): 1521–1528. doi: 10.1021/ef9011005 DOI: https://doi.org/10.1021/ef9011005
40. Arutyunov VS, Strekova LN, Savchenko VI, et al. Prospects of Conversion of Hydrocarbon Gases to Liquid Products Based on Nitrogen-Rich Synthesis Gas (Review). Petroleum Chemistry. 2019; 59(4): 370–379. doi: 10.1134/S0965544119040029 DOI: https://doi.org/10.1134/S0965544119040029
41. Hedden K, Jess A, Kuntze T. From Natural gas to liquid hydrocarbons (1). A new concept for the production of liquid hydrocarbons from natural gas in remote areas. OIL GAS – European Magazine. 1994; 3: 42–44.
42. Jess A, Popp R, Hedden K. Fischer–Tropsch-synthesis with nitrogen-rich syngas. Applied Catalysis A: General. 1999; 186(1–2): 321–342. doi: 10.1016/S0926-860X(99)00152-0 DOI: https://doi.org/10.1016/S0926-860X(99)00152-0
43. Kertthong T, Schmid M, Scheffknecht G. Non-catalytic partial oxidation of methane in biomass-derived syngas with high steam and hydrogen content optimal for subsequent synthesis process. Journal of the Energy Institute. 2022; 105: 251–261. doi: 10.1016/j.joei.2022.09.007 DOI: https://doi.org/10.1016/j.joei.2022.09.007
44. Arutyunov VS, Nikitin AV, Savchenko VI, et al. Combined Production of Hydrogen and Methanol without CO2 Emission Based on Matrix Conversion of Natural Gas. Doklady Chemistry. 2023; 513(2): 361–366. doi: 10.1134/S0012500823601018 DOI: https://doi.org/10.1134/S0012500823601018
45. JSC “TOU”. Electrochemical generator on solid oxide fuel cells. Patent RU 2608749 С1, 24 January 2017.
46. Arutyunov VS, Shmelev VM, Rakhmetov AN, et al. New approaches to development of low-emission combustion chambers for gas turbine engines. International Scientific Journal for Alternative Energy and Ecology. 2013; 6 (128): 105–120. (in Russian)
47. Lyalin DA, Vasilik NY, Zaharov AA, et al. Low-emission efficient infrared burner for cooking applications. Oil & Gas Chemistry. 2018; 4: 5–9. doi: 10.24411/2310-8266-2018-10401 (in Russian)
48. Qazi UY. Future of Hydrogen as an Alternative Fuel for Next-Generation Industrial Applications; Challenges and Expected Opportunities. Energies. 2022; 15(13): 4741. doi: 10.3390/en15134741 DOI: https://doi.org/10.3390/en15134741
49. Lamari F, Weinberger B, Langlois P, et al. Instances of Safety-Related Advances in Hydrogen as Regards Its Gaseous Transport and Buffer Storage and Its Solid-State Storage. Hydrogen. 2024; 5(3): 387–402. doi: 10.3390/hydrogen5030022 DOI: https://doi.org/10.3390/hydrogen5030022




.jpg)
.jpg)

