Thermal Decomposition Behavior and Kinetic Analysis of Lithium-Ion Battery Electrolyte under Different Atmospheric Conditions
DOI:
https://doi.org/10.18686/cest594Keywords:
electrolyte , thermal decomposition , coats–redfern methodAbstract
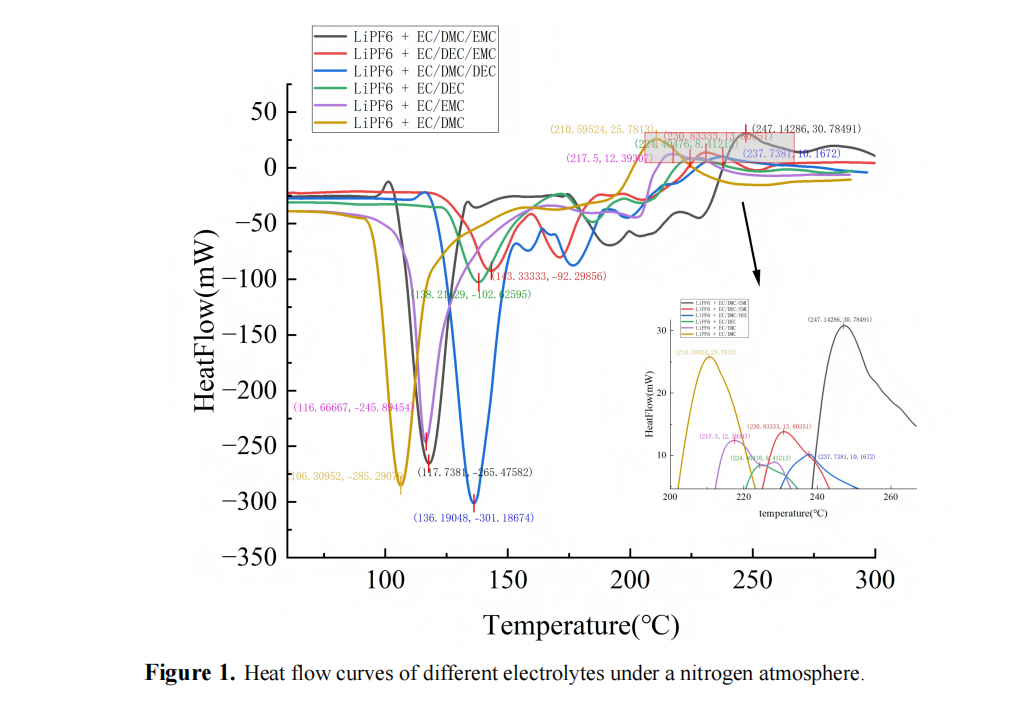
To gain a deeper understanding of the thermal reaction behavior and thermal safety characteristics of lithium-ion battery electrolytes under different atmospheric conditions, this study systematically investigates the thermal decomposition process of 1 mol·L⁻¹ LiPF6 dissolved in three typical solvent systems—EC/EMC, EC/EMC/DMC, and EC/EMC/DEC—using a high-precision microcalorimeter (C600). The analysis is performed under two typical atmospheric conditions: closed adiabatic and nitrogen flow. The heat flow and differential heat flow curves are quantitatively analyzed, and the exothermic onset temperature, thermal intensity, and reaction complexity are compared by integrating the cumulative heat release enthalpy. Furthermore, the Coats–Redfern model is used to extract kinetic parameters, perform global and α-segment fitting, and analyze the trend of reaction path changes. The results show that nitrogen flow significantly inhibits chain-side reactions and improves thermal stability, while under closed conditions, overlapping exothermic peaks and multi-stage releases are more likely to occur. Kinetic analysis reveals that the EC/EMC system exhibits a sharp increase in activation energy during the high conversion stage, suggesting a higher potential for thermal runaway, whereas the EC/EMC/DMC system has a single reaction mechanism and higher activation energy, indicating better thermal safety.
Downloads
Published
How to Cite
Issue
Section
License
Copyright (c) 2026 Rongkun Pan, Jiayi Yu, Jiangkun Chao, Daimin Hu

This work is licensed under a Creative Commons Attribution 4.0 International License.
References
1. Balakrishnan PG, Ramesh R, Kumar TP. Safety mechanisms in lithium-ion batteries. Journal of Power Sources. 2006; 155(2): 401–414. doi: 10.1016/j.jpowsour.2005.12.002 DOI: https://doi.org/10.1016/j.jpowsour.2005.12.002
2. Lyon RE, Walters RN. Energetics of lithium ion battery failure. Journal of Hazardous Materials. 2016; 318: 164–172. doi: 10.1016/j.jhazmat.2016.06.047 DOI: https://doi.org/10.1016/j.jhazmat.2016.06.047
3. Fernandes Y, Bry A, de Persis S. Thermal degradation analyses of carbonate solvents used in Li-ion batteries. Journal of Power Sources. 2019; 414: 250–261. doi: 10.1016/j.jpowsour.2018.12.077 DOI: https://doi.org/10.1016/j.jpowsour.2018.12.077
4. Liu L, Shadike Z, Wang N, et al. Low concentration electrolyte: A new approach for achieving high performance lithium batteries. eScience. 2024; 4(6): 100268. doi: 10.1016/j.esci.2024.100268 DOI: https://doi.org/10.1016/j.esci.2024.100268
5. Cresce AV, Xu K. Aqueous lithium-ion batteries. Carbon Energy. 2021; 3(5): 721–751. doi: 10.1002/cey2.106 DOI: https://doi.org/10.1002/cey2.106
6. Yan Y. Development Status of Lithium-ion Battery Electrolytes Based on LiPF6. Coal and Chemical Industry. 2013; 36(9): 28–31+41. (in Chinese)
7. Campion CL, Li W, Lucht BL. Thermal Decomposition of LiPF6-Based Electrolytes for Lithium-Ion Batteries. Journal of The Electrochemical Society. 2005; 152(12): A2327. doi: 10.1149/1.2083267 DOI: https://doi.org/10.1149/1.2083267
8. Said MSM, Tohir MZM. Characterisation of thermal runaway behaviour of cylindrical lithium-ion battery using Accelerating Rate Calorimeter and oven heating. Case Studies in Thermal Engineering. 2021; 28: 101474. doi: 10.1016/j.csite.2021.101474 DOI: https://doi.org/10.1016/j.csite.2021.101474
9. Ren D, Liu X, Feng X, et al. Model-based thermal runaway prediction of lithium-ion batteries from kinetics analysis of cell components. Applied Energy. 2018; 228: 633–644. doi: 10.1016/j.apenergy.2018.06.126 DOI: https://doi.org/10.1016/j.apenergy.2018.06.126
10. Wu T, Chen H, Wang Q, et al. Comparison analysis on the thermal runaway of lithium-ion battery under two heating modes. Journal of Hazardous Materials. 2018; 344: 733–741. doi: 10.1016/j.jhazmat.2017.11.022 DOI: https://doi.org/10.1016/j.jhazmat.2017.11.022
11. Yeh FM, Volli V, Bin LW, et al. Oxidative stability and thermal performance of ester based lube oil with lithium salt additives. Applied Thermal Engineering. 2019; 150: 1328–1336. doi: 10.1016/j.applthermaleng.2019.01.061 DOI: https://doi.org/10.1016/j.applthermaleng.2019.01.061
12. Lamb J, Orendorff CJ, Roth EP, et al. Studies on the Thermal Breakdown of Common Li-Ion Battery Electrolyte Components. Journal of The Electrochemical Society. 2015; 162(10): A2131. doi: 10.1149/2.0651510jes DOI: https://doi.org/10.1149/2.0651510jes
13. Liao Z, Zhang S, Zhao Y, et al. Experimental evaluation of thermolysis-driven gas emissions from LiPF6-carbonate electrolyte used in lithium-ion batteries. Journal of Energy Chemistry. 2020; 49(10): 124–135. doi: 10.1016/j.jechem.2020.01.030 DOI: https://doi.org/10.1016/j.jechem.2020.01.030
14. Tian Y, Zhan X, Zhang Y, et al. Thermal Decomposition Mechanism of PF5 and POF3 with Carbonate-Based Electrolytes During Lithium-Ion Batteries' Thermal Runaway. Fire. 2025; 8(9): 370. doi: 10.3390/fire8090370 DOI: https://doi.org/10.3390/fire8090370
15. Salomez B, Grugeon S, Tran-Van P, et al. Counteracting thermal degradation of LiPF6-based electrolyte with additives or lithium salts: A gas analysis revealing the impact of NMC. Journal of Power Sources. 2024; 613: 234901. doi: 10.1016/j.jpowsour.2024.234901 DOI: https://doi.org/10.1016/j.jpowsour.2024.234901
16. Yin J, Cui S, Mo W, et al. Kinetics of Isothermal Phase Transformation Kinetics of U–2Nb Alloy. Rare Metal Materials and Engineering. 2022; 51(12): 4508–4518. doi: 10.12442/j.issn.1002-185X.20211151
17. Bondarchuk I, Bondarchuk S, Vorozhtsov A, et al. Advanced Fitting Method for the Kinetic Analysis of Thermogravimetric Data. Molecules. 2023; 28(1): 424. doi: 10.3390/molecules28010424 DOI: https://doi.org/10.3390/molecules28010424
18. Sait HH, Hussain A, Salema AA, et al. Pyrolysis and combustion kinetics of date palm biomass using thermogravimetric analysis. Bioresource Technology. 2012; 118: 382–389. doi: 10.1016/j.biortech.2012.04.081 DOI: https://doi.org/10.1016/j.biortech.2012.04.081
19. Krishnan G, Rajesh K, Krishnan P, et al. A study on kinetic properties of brucinium hydrogen(s) malate pentahydrate single crystal by Coats Redfern method. AIP Conference Proceedings. 2020; 2265(1), 030425. doi: 10.1063/5.0017481 DOI: https://doi.org/10.1063/5.0017481
20. Naqvi SR, Uemura Y, Osman N, et al. Kinetic study of the catalytic pyrolysis of paddy husk by use of thermogravimetric data and the Coats-Redfern model. Research on Chemical Intermediates. 2015; 41(12): 9743–9755. doi: 10.1007/s11164-015-1962-0 DOI: https://doi.org/10.1007/s11164-015-1962-0
21. Han KS, Lee MS, Kim N, et al. Lithium-ion hopping weakens thermal stability of LiPF6 carbonate electrolytes. Cell Reports Physical Science. 2024; 5(1): 101768. doi: 10.1016/j.xcrp.2023.101768 DOI: https://doi.org/10.1016/j.xcrp.2023.101768
22. Kawamura T, Kimura A, Egashira M, et al. Thermal stability of alkyl carbonate mixed-solvent electrolytes for lithium ion cells. Journal of Power Sources. 2002; 104: 260–264. doi: 10.1016/S0378-7753(01)00960-0 DOI: https://doi.org/10.1016/S0378-7753(01)00960-0
23. Du G, Zheng L, Zhang Z, et al. Overview of research on thermal safety of lithium-ion batteries. Energy Storage Science and Technology. 2019; 8(3): 500–505. (in Chinese)
24. Wang JB, Wang XB, Li XB. Progress in research on improving the safety of lithium-ion battery electrolytes. Fire Science and Technology. 2025; 44(1): 41–47. doi: 10.20168/j.1009-0029.2025.01.0041.07 (in Chinese)
25. Zhang GD. Study on Gas Generation Characteristics during Thermal Runaway of Lithium-ion Battery Electrolytes [Master's Thesis]. Northeastern University; 2022. doi: 10.27007/d.cnki.gdbeu.2022.002476 (in Chinese)




.jpg)
.jpg)

