Comparison of oxy-fuel combustion kinetic mechanisms of methane by laminar burning velocity under normal conditions
DOI:
https://doi.org/10.18686/cest604Keywords:
oxy-fuel combustion; normal conditions; laminar burning velocity; verification of kinetic mechanisms; UoS sCO2; OXY-NG; GRI; SkeletalAbstract
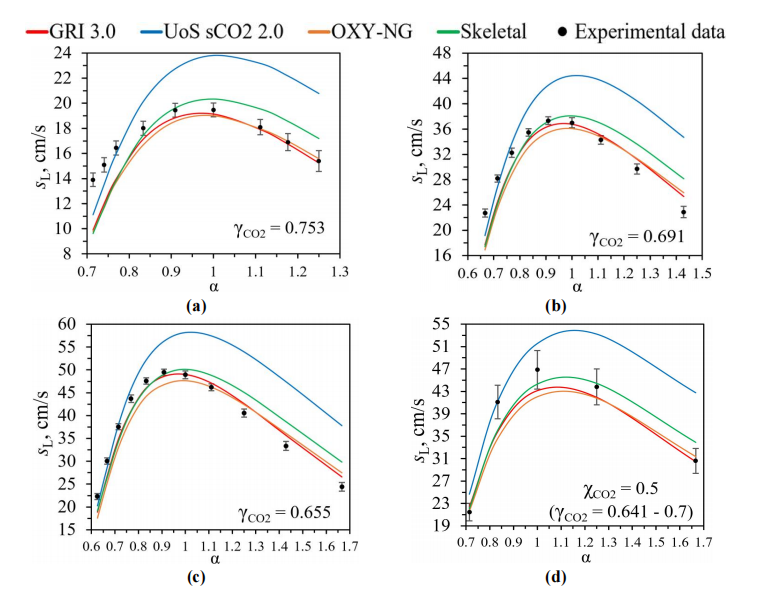
Power energy sector is the largest anthropogenic source of greenhouse gas emissions, including CO2. Oxy-fuel energy complexes (OFCs) are one of the most effective ways to reduce CO2 emissions. In OFC’s combustion chamber gaseous fuel is burned in a mixture of O2 and CO2 at supercritical pressures up to 300 atm. However, in open sources there are currently no recommendations for designing such combustion chambers, including no recommendations on the choice of a kinetic mechanism for numerical simulation of combustion. In this paper, the kinetic combustion mechanisms GRI 3.0, UoS sCO2 2.0, OXY-NG, and Skeletal were compared using Chemkin 18.2 for oxy-fuel combustion of methane by laminar burning velocity under normal conditions. It is shown that OXY-NG can accurately simulate the laminar burning velocity during oxy-fuel combustion of methane under normal conditions in a wide range of mixture compositions by (α) 0.8–1.4 and (γCO2) 0.65–0.78. For this reason, OXY-NG mechanism is recommended for spatial numerical simulation of oxy-fuel combustion.
Downloads
Published
How to Cite
Issue
Section
License
Copyright (c) 2026 Andrey Rogalev, Sergey Osipov, Vadim Yakovlev, Maxim Kozhemiakin, Dmitry Pisarev

This work is licensed under a Creative Commons Attribution 4.0 International License.
References
1. US EPA OAR, Global Greenhouse Gas Overview. Available online: https://www.epa.gov/ghgemissions/global-greenhouse-gas-overview (accessed on 6 October 2025).
2. Jansen D, Gazzani M, Manzolini G, et al. Pre-combustion CO2 capture. International Journal of Greenhouse Gas Control. 2015; 40: 167–187. doi: 10.1016/j.ijggc.2015.05.028 DOI: https://doi.org/10.1016/j.ijggc.2015.05.028
3. Allam R, Martin S, Forrest B, et al. Demonstration of the Allam cycle: An update on the development status of a high efficiency supercritical carbon dioxide power process employing full carbon capture. Energy Procedia. 2017; 114: 5948–5966. doi: 10.1016/j.egypro.2017.03.1731 DOI: https://doi.org/10.1016/j.egypro.2017.03.1731
4. Kindra VO. Improving the efficiency of oxy-fuel power systems with carbon dioxide working fluid using structural and parametric optimization of thermal circuits. Cand. Moscow Power Engineering Institute, Moscow; 2019.
5. Cision. NET Power Demonstration Plant Wins 2018 ADIPEC Breakthrough Technological Project of the Year. Available online: https://www.prnewswire.com/news-releases/net-power-demonstration-plant-wins-2018-adipec-breakthrough-technological-project-of-the-year-300750163.html. (accessed on 28 November 2025).
6. Osipov S, Sokolov V, Yakovlev V, et al. Comparative analysis of the oxy-fuel kinetic mechanisms by the ignition delay time of methane. Energies. 2025; 18(9): 2155. doi: 10.3390/en18092155 DOI: https://doi.org/10.3390/en18092155
7. Harman-Thomas JM, Hughes KJ, Pourkashanian M. The development of a chemical kinetic mechanism for combustion in supercritical carbon dioxide. Energy. 2022; 255: 124490. doi: 10.1016/j.energy.2022.124490 DOI: https://doi.org/10.1016/j.energy.2022.124490
8. Harman-Thomas JM, Kashif TA, Hughes KJ, et al. Experimental and modelling study of hydrogen ignition in CO2 bath gas. Fuel. 2023; 334: 126664. doi: 10.1016/j.fuel.2022.126664 DOI: https://doi.org/10.1016/j.fuel.2022.126664
9. Harman-Thomas JM, Kashif TA, Hughes KJ, et al. Experimental and modelling study of syngas combustion in CO2 bath gas. Fuel. 2023; 342: 127865. doi: 10.1016/j.fuel.2023.127865 DOI: https://doi.org/10.1016/j.fuel.2023.127865
10. Harman-Thomas JM, Kashif TA, Hughes KJ, et al. Autoignition of methane and methane/hydrogen blends in CO2 bath gas. Fuel. 2025; 381. DOI: https://doi.org/10.1016/j.fuel.2024.133229
11. Harman‐Thomas JM, Ingham DB, Hughes KJ, et al. Role of methyldioxy radical chemistry in high‐pressure methane combustion in CO2. International Journal of Chemical Kinetics. 2023; 55(10): 629–641. doi: 10.1002/kin.21672 DOI: https://doi.org/10.1002/kin.21672
12. Liu Y, Zou C, Cheng J, et al. Experimental and numerical study of the effect of CO2 on the ignition delay times of methane under different pressures and temperatures. Energy & Fuels. 2018; 32(10): 10999–11009. doi: 10.1021/ DOI: https://doi.org/10.1021/acs.energyfuels.8b02443
13. acs.energyfuels.8b02443
14. Liu Y, Cheng J, Zou C, et al. Ignition delay times of ethane under O2/CO2 atmosphere at different pressures by shock tube and simulation methods. Combustion and Flame. 2019; 204: 380–390. doi: 10.1016/j.combustflame.2019.03.031 DOI: https://doi.org/10.1016/j.combustflame.2019.03.031
15. Xia W, Peng C, Zou C, et al. Shock tube and modeling study of ignition delay times of propane under O2/CO2/Ar atmosphere. Combustion and Flame. 2020; 220: 34–48. doi: 10.1016/j.combustflame.2020.06.024 DOI: https://doi.org/10.1016/j.combustflame.2020.06.024
16. Xia W, Zou C, Yuan Y, et al. Experimental and numerical study on the ignition delay times of methane/propane mixtures diluted in carbon dioxide. Fuel. 2023; 343: 127991. doi: 10.1016/j.fuel.2023.127991 DOI: https://doi.org/10.1016/j.fuel.2023.127991
17. Xia W, Huang C, Yang J, et al. Experimental and modeling study of ignition delay times of natural gas with CO2 dilution. Fuel. 2024; 358: 130148. doi: 10.1016/j.fuel.2023.130148 DOI: https://doi.org/10.1016/j.fuel.2023.130148
18. Smith GP, Golden DM, Frenklach M, et al. GRI mechanism 3.0 (1999). Available online: http://combustion.
19. berkeley.edu/gri-mech/version30/text30.html (accessed on 5 October 2025).
20. Hu F, Li P, Guo J, et al. Evaluation, development, and validation of a new reduced mechanism for methane oxy-fuel combustion. International Journal of Greenhouse Gas Control. 2018; 78: 327–340. doi: 10.1016/j.ijggc.2018.08.018 DOI: https://doi.org/10.1016/j.ijggc.2018.08.018
21. Wang S, Wang Z, He Y, et al. Laminar burning velocities of CH4/O2/N2 and oxygen-enriched CH4/O2/CO2 flames at elevated pressures measured using the heat flux method. Fuel. 2020; 259: 116152. doi: 10.1016/j.fuel.2019.116152 DOI: https://doi.org/10.1016/j.fuel.2019.116152
22. Khan AR, Anbusaravanan S, Kalathi L, et al. Investigation of dilution effect with N2/CO2 on laminar burning velocity of premixed methane/oxygen mixtures using freely expanding spherical flames. Fuel. 2017; 196: 225–232. doi: 10.1016/j.fuel.2017.01.086 DOI: https://doi.org/10.1016/j.fuel.2017.01.086
23. Han X, Ling Z, Zhang G, et al. Experimental and modelling study on the laminar burning velocity correlation of CH4 flames diluted by different diluents. Fuel. 2024; 370: 131860. doi: 10.1016/j.fuel.2024.131860 DOI: https://doi.org/10.1016/j.fuel.2024.131860
24. Hu X, Yu Q, Liu J, et al. Investigation of laminar flame speeds of CH4/O2/CO2 mixtures at ordinary pressure and kinetic simulation. Energy. 2014; 70: 626-634. doi: 10.1016/j.energy.2014.04.029 DOI: https://doi.org/10.1016/j.energy.2014.04.029




.jpg)
.jpg)

